- Blog
- Crack fuel pc game
- Watch dragon ball z broly the legendary super saiyan movie
- Trainer gta 4 1-0-7-0
- My hero academia season 3 reddit ep 13
- Sonic unleashed project
- Launch a phone dialer app android studio
- Adobe acrobat 7-0 professional activation code free download
- Microsoft encarta 2013 torrent
- Softorbits photo stamp remover 8-4 torrent
- Phan mem video edit magic 4-47 crack
- When can you download the sims 4 cats and dogs preorder
- The strange story of a guy next door and a novelist raws
- Download viki videos with subs
- Blinded by the light bruce
- Gta v disc 2 iso download xbox 360 usa
- Sades 7-1 not working
- Block in ufc 3
- Modern physical organic chemistry sar experiment
- Splinter cell conviction torrent
- Drivers para camara web genius eye 312
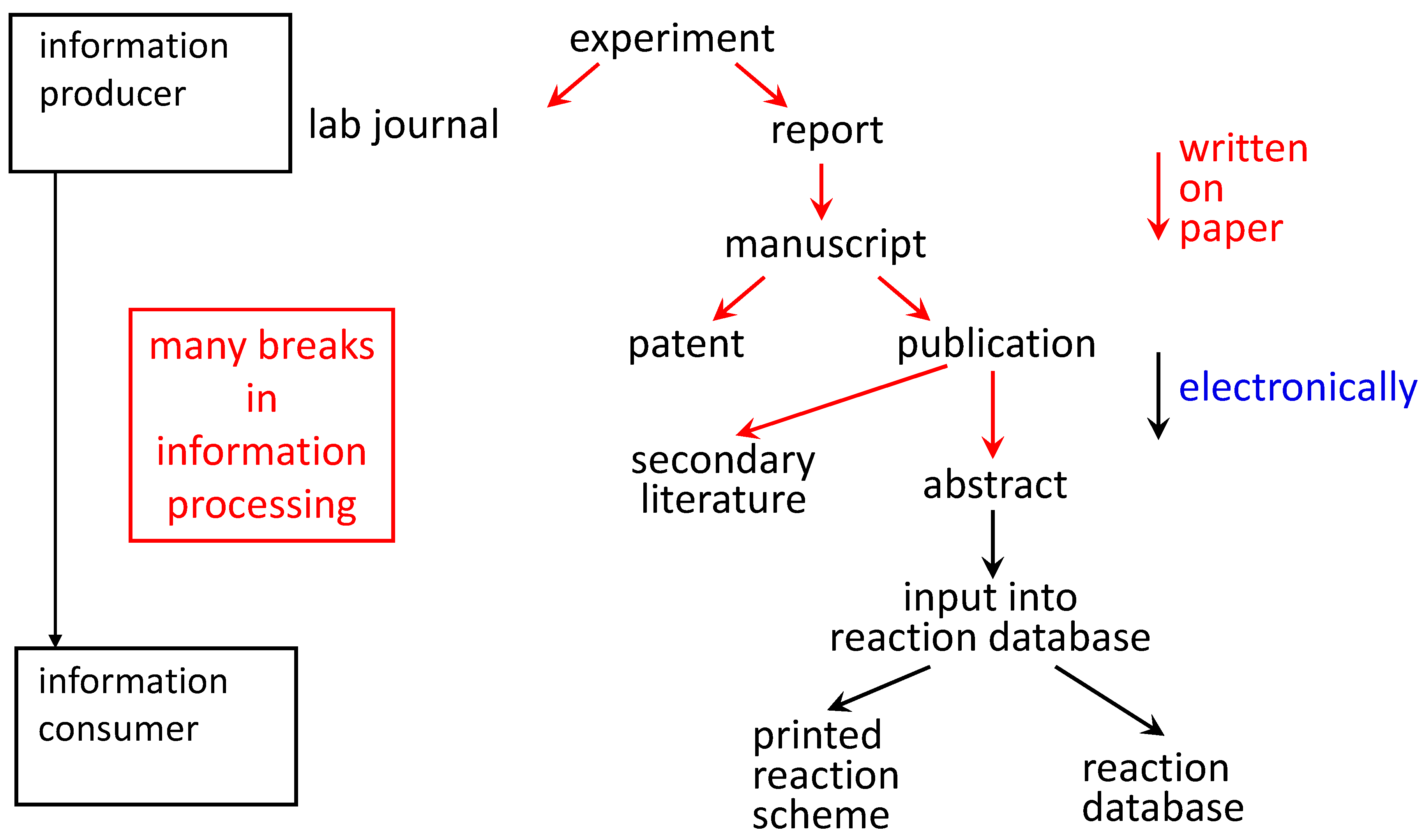
Experiments investigate the quantum behavior of atoms and molecules, thermodynamics, and equilibria. Prerequisites: Chemistry 2 or 12, Mathematics 34 or equivalent, and Physics 2 or 12, or consent.

Applications of the principles of modern physical chemistry to problems in chemical bonding, atomic and molecular structure, spectroscopy, and chemical kinetics. Physics may be taken concurrently.Ġ032 Physical Chemistry II. Only one of Chemistry 1, 11, or 16 may be counted for credit.Ġ031 Physical Chemistry I. Introduction to the principles of modern physical chemistry:elementary wave mechanics, atomic structure, and chemical bonding, elementary statistical thermodynamics, and the thermodynamic basis for phase behavior, chemical reactivity and equilibrium in gases and liquid solutions. Prerequisite: Good background in mathematics. Three lectures, one recitation, one laboratory. This one-semester course may be used in conjunction with Chemistry 2 to fulfill the basic chemistry requirement for a chemistry major. Topics include atomic and molecular structure, intermolecular forces, ionic and covalent bonding.
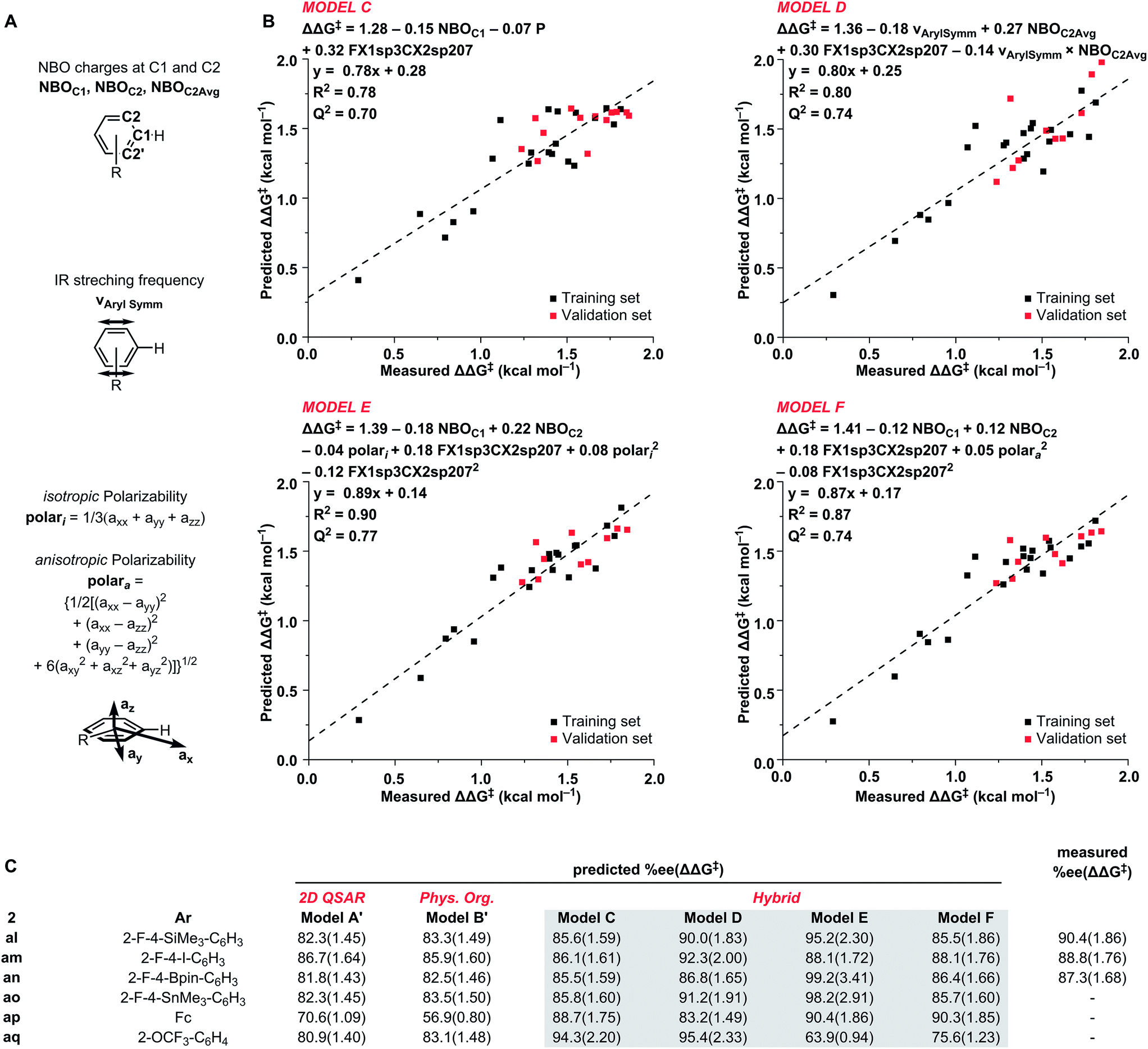
An introductory course investigating the fundamentals and principles of chemistry through exploration of modern materials, e.g., thin films, superconductors, ultrasmall structures, modern electronics and photonics. Only one of Chemistry 1, 11, or 16 and one of Chemistry 2 or 12 may be counted for credit.Ġ016 Chemistry of Materials. Prerequisites: Score of at least 4 on the AP chemistry exam or consent Mathematics 32 (may be taken concurrently). Three lectures, one seminar on frontiers in chemistry, one laboratory, one recitation. Some familiarity with elementary calculus concepts assumed. For well-prepared students intending to be science majors. Designed to provide a strong foundation for advanced courses in chemistry. Topics covered are the same as in Chemistry 1 and 2, but discussed in greater detail and with a higher degree of mathematical rigor.

Spring 2000 and alternate years.Ġ011, 0012 General Chemistry. The concept of equilibrium in complex systems thermodynamic limits and kinetic realities. An introductory course designed primarily to give non-science majors an appreciation of basic chemical principles underlying the causes of and possible solutions to current environmental problems. This course does not fulfill pre-medical requirements for a lab-based chemistry course.Ġ008 Environmental Chemistry. Students will learn the evidence for the various ideas presented, the scientific method used by scientists, and how the community of scientists evaluates the evidence. Only one of Chemistry 2 or 12 may be counted for credit.Ġ006 From the Big Bang to Humankind. Course will explore the origins of the Universe, the formation of Earth and its structure, the chemistry of life, the development of complex organisms, and the development of modern humans. Prerequisite: Chemistry 1, 11, 16, or consent. Three lectures, one laboratory, one recitation. Additional topics may include environmental, nuclear, coordination chemistry chemistry of selected elements and introduction to biological chemistry. Only one of Chemistry 1, 11, or 16, may be counted for credit.Ġ002 Chemical Principles with Lab. Chemical kinetics and thermodynamics, physical and chemical equilibria, aqueous equilibria (acid-base, precipitation, and complex formation), electrochemistry, introduction to organic chemistry (families of organic compounds, basic stereochemistry and nomenclature). Atomic and molecular structure, chemical nomenclature, intermolecular forces and states of matter, the relation of structure and bonding to physical and chemical properties of matter, patterns of chemical reactions, stoichiometry, thermochemistry, and properties of solutions. Undergraduate Only (CHEM 0001-0092)Ġ001 Chemical Fundamentals with Lab. For up-to-date information on course offerings, schedules, room locations and registration, please visit the Student Information System (SIS). Visit the undergraduate and graduate pages for course requirements for specific programs. The list below includes descriptions of all undergraduate and graduate courses offered by the Department of Chemistry, though some courses may be taught more often than others.
- Blog
- Crack fuel pc game
- Watch dragon ball z broly the legendary super saiyan movie
- Trainer gta 4 1-0-7-0
- My hero academia season 3 reddit ep 13
- Sonic unleashed project
- Launch a phone dialer app android studio
- Adobe acrobat 7-0 professional activation code free download
- Microsoft encarta 2013 torrent
- Softorbits photo stamp remover 8-4 torrent
- Phan mem video edit magic 4-47 crack
- When can you download the sims 4 cats and dogs preorder
- The strange story of a guy next door and a novelist raws
- Download viki videos with subs
- Blinded by the light bruce
- Gta v disc 2 iso download xbox 360 usa
- Sades 7-1 not working
- Block in ufc 3
- Modern physical organic chemistry sar experiment
- Splinter cell conviction torrent
- Drivers para camara web genius eye 312
